If you've ever looked into how generic drugs are approved, you might have stumbled across a confusing statistic: the "80-125% rule." At first glance, it sounds alarming. You might think it means your generic pill could have 25% more or 20% less active medicine than the brand-name version. If that were true, your dose would be wildly inconsistent. But here is the reality: that is a complete myth. The rule isn't about how much powder is in the tablet; it's about how the drug behaves once it's inside your body. Understanding this distinction is the key to trusting the 80-125% rule and knowing why your generic medication works just as well as the expensive brand.
What is Bioequivalence Actually Measuring?
To understand the rule, we first have to define Bioequivalence is the absence of a significant difference in the rate and extent to which the active ingredient becomes available at the site of drug action . When you swallow a pill, it doesn't just vanish into your bloodstream. It has to dissolve, pass through the gut wall, and survive the liver before it hits your target organ. This process is called absorption.
The FDA doesn't just weigh the active ingredient in a lab; they track the drug's journey in humans. They look at two main things: how much of the drug gets absorbed (the extent) and how fast it gets there (the rate). If a generic drug delivers the active ingredient to your bloodstream in a way that is statistically indistinguishable from the brand name, it's considered bioequivalent. This means the patient gets the same clinical effect, regardless of which version they take.
Breaking Down the 80-125% Range
The numbers 80% and 125% aren't random, and they aren't "allowances" for mistakes in manufacturing. They are part of a statistical calculation. Because human bodies are all different-some of us absorb drugs faster or slower than others-there is always a bit of "noise" in the data. To account for this, scientists use a 90% confidence interval (CI).
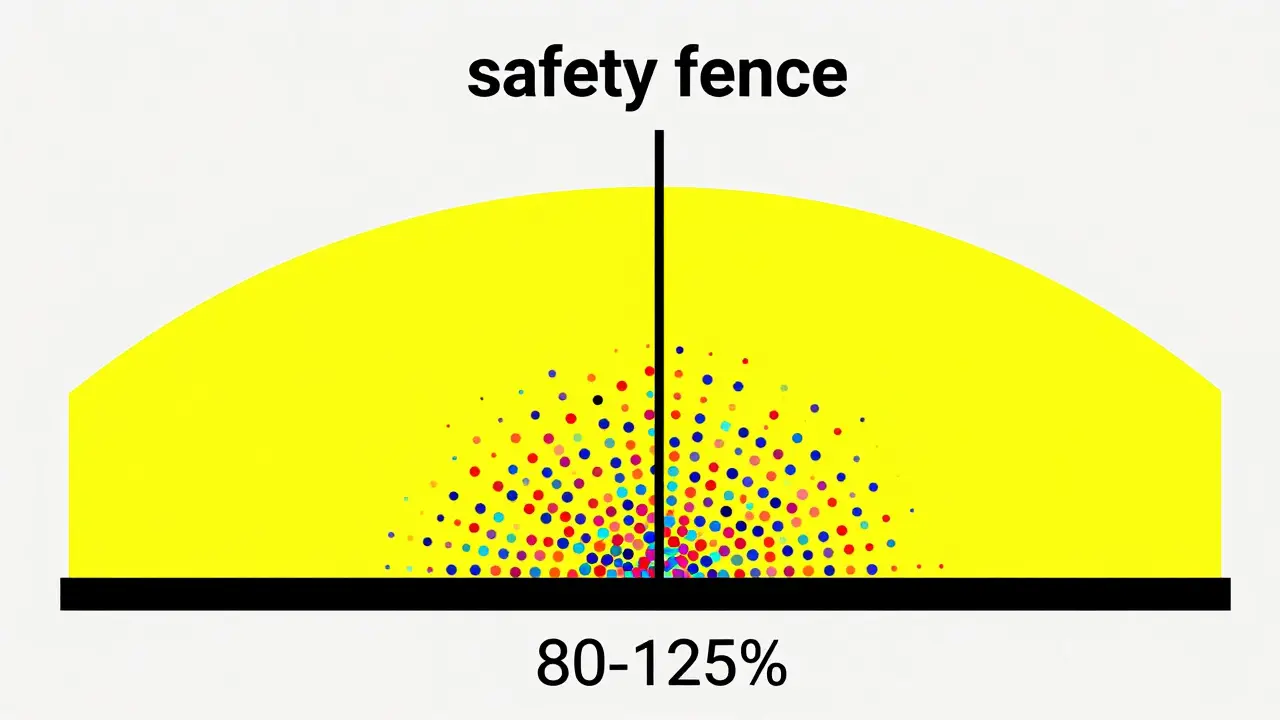
For a generic to pass, the entire 90% confidence interval of the ratio between the generic and the brand must fall within the 80-125% range. This is a much tougher test than it sounds. If the average absorption of a generic is slightly off, the "tails" of that confidence interval will likely dip below 80% or jump above 125%, and the drug will be rejected. In fact, FDA data shows that 98% of approved generics actually have an average absorption within 95-105% of the brand name. The 80-125% window is the "safety fence," but most generics are sitting almost exactly on the 100% line.
| Metric | What it Measures | Why it Matters |
|---|---|---|
| AUC (Area Under the Curve) | Total systemic exposure | Ensures the total amount of drug reaching the blood is correct. |
| Cmax (Maximum Concentration) | Peak plasma concentration | Ensures the drug doesn't hit the system too fast (toxicity) or too slow (ineffectiveness). |
The Math Behind the Asymmetry
You might wonder why it's 125% and not 120%. If the bottom limit is 80% (a 20% drop), why isn't the top limit 120% (a 20% rise)? This comes down to how scientists handle data. In pharmacokinetics, data is usually "log-normally distributed." This means scientists take the natural logarithm of the numbers to make the data symmetrical for analysis.
When you convert a 20% decrease (0.80) into a logarithm, you get approximately -0.223. To keep the math balanced, if you add that same 0.223 to the center point (log of 1.0), and then convert it back to a normal number, you get 1.25. So, 80-125% is actually a perfectly symmetrical $\pm$20% range on a logarithmic scale. It's not a "bonus" for the generic company; it's just how the math works.
When the Standard Rules Aren't Enough
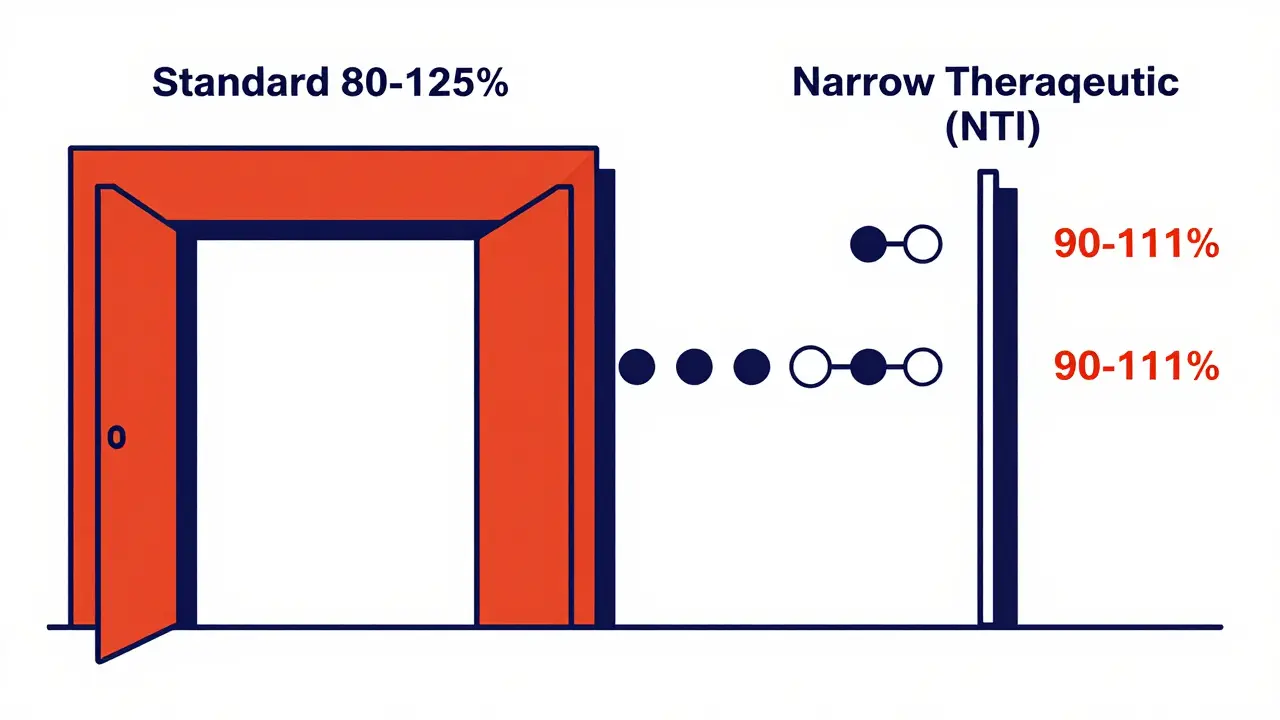
While the 80-125% rule works for most medications, some drugs are far more sensitive. These are called Narrow Therapeutic Index (NTI) drugs, which are medications where a tiny change in dose can be the difference between a cure and a toxic reaction. Examples include warfarin (a blood thinner) and levothyroxine (for thyroid health).
For these high-stakes medications, the FDA uses much tighter boundaries. Instead of the standard range, they often require a window of 90-111%. This ensures that the generic is almost an exact mirror of the brand-name drug, leaving virtually no room for variation. This tiered approach shows that the 80-125% rule isn't a "one size fits all" guess, but a calculated standard based on the drug's risk profile.
Real-World Evidence: Does it Actually Work?
If you're still skeptical, look at the data. A massive study published in JAMA Internal Medicine analyzed 2 million patient records and found that for cardiovascular drugs, there was no meaningful difference in clinical outcomes between those taking brands and those taking generics. Similarly, the FDA's Sentinel Initiative, which monitors hundreds of millions of records, found no significant difference in adverse event rates for 94% of evaluated products.
The impact of these standards is seen in the economy as well. Generics now make up about 90% of all prescriptions in the U.S. If the 80-125% rule were actually causing drugs to be 20% weaker or stronger, we would see a massive spike in hospitalizations. Instead, we see billions of dollars in healthcare savings and stable patient outcomes. The rule allows for natural human biological variation while guaranteeing that the medicine does its job.
Common Pitfalls and Misunderstandings
The biggest mistake people make is confusing "bioequivalence" with "chemical purity." A generic drug must contain the same amount of active ingredient as the brand name-usually within a very tight percentage (typically 95-105% of the label claim). The 80-125% rule describes how that drug is absorbed into the blood, not how much is in the pill.
Another common concern is the use of different "inactive ingredients" (excipients). While the active drug is the same, generics use different fillers, binders, or dyes. Some people believe these change the absorption rate. While this is why the bioequivalence tests are performed in the first place, the 80-125% test specifically proves that these different fillers do not significantly interfere with how the drug enters your system.
Does 80-125% mean my generic drug is 20% weaker?
No. The 80-125% range refers to the statistical confidence interval of the absorption rate, not the amount of active ingredient. Most approved generics have an average absorption within 5% of the brand-name drug.
Who decides if a drug is bioequivalent?
In the United States, the FDA (Food and Drug Administration) sets the standards and reviews the bioequivalence studies. Similar agencies like the EMA in Europe follow nearly identical rules.
Why can't the rule be exactly 100%?
Because humans are biologically different. Two different people taking the exact same brand-name pill will have different absorption rates. The 80-125% range accounts for this natural human variability.
Are there any drugs where generics aren't recommended?
For most people, generics are interchangeable. However, for Narrow Therapeutic Index (NTI) drugs, some doctors prefer patients stay on one consistent version (either brand or a specific generic) to avoid even tiny fluctuations in blood levels.
What happens if a generic fails the 80-125% test?
The FDA will not approve the drug for sale. The manufacturer must either change the formulation to improve absorption or provide additional data to prove the drug is still therapeutically equivalent.
Next Steps for Patients and Providers
If you are a patient worried about your medication, the best move is to ask your pharmacist for the specific generic manufacturer. While all approved generics meet the 80-125% rule, staying with the same manufacturer can provide extra peace of mind regarding consistency.
For healthcare providers, educating patients about the "logarithmic scale" and the difference between active ingredient weight and blood absorption can alleviate a lot of anxiety. Using the term "therapeutic equivalence" instead of "identical" helps set realistic expectations: the drugs aren't clones in every single way (like color or shape), but they are clones in the way they treat the disease.